What is Water?
Water is made up of two hydrogen atoms and one oxygen atom (H2O). An atom is the smallest unit of matter that retains its properties. Atoms consists of protons, electrons, and neutrons. Atoms are usually neutrually charged because the number of protons and electrons are even. When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen, and the oxygen atom shares one of its electrons with the hydrogen atom.
Water is made up of two hydrogen atoms and one oxygen atom (H2O). An atom is the smallest unit of matter that retains its properties. Atoms consists of protons, electrons, and neutrons. Atoms are usually neutrually charged because the number of protons and electrons are even. When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen, and the oxygen atom shares one of its electrons with the hydrogen atom.
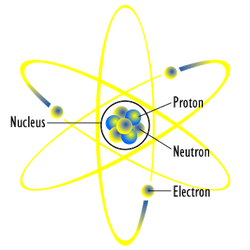
Protons: have a positive charge .
Electrons: have a negative charge .
Neutrons: have no charge (neutral).
Ions: unevenly charged atoms.
Electrons: have a negative charge .
Neutrons: have no charge (neutral).
Ions: unevenly charged atoms.
Water is a Polar Molecule:
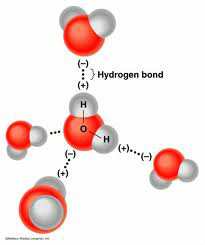
Shared electrons spend more time around the oxygen atom than the hydrogen atom. The hydrogen atom carries a slightly positive charge while the oxygen atom carries a slightly negative charge. In other words, the oxygen atom is negative and the hydrogen atom is positive. Because of that, the oxygen atom and the hydrogen atoms are attracted to each other.
Hydrogen Bonding is a weak attraction between the weakly positive hydrogen atoms and the weakly negative oxygen atoms of a different molecule. Hydrogen bonding is responsible for the unique properties of water. That's mean they all gather together to make water.
Hydrogen Bonding is a weak attraction between the weakly positive hydrogen atoms and the weakly negative oxygen atoms of a different molecule. Hydrogen bonding is responsible for the unique properties of water. That's mean they all gather together to make water.
Water is a Universal Solvent:
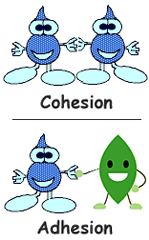
Polarity of water gives it the ability to dissolve most compounds. The positive hydrogen side of water is attracted to the negative parts of the compound it is dissolving. Negative oxygen is attracted to the positive parts. This allows water to dissolve and break apart ionically charged substances. Cohesion is when water is attracted to other water molecules. This helps trees pull water high up into their branches. Water is able to pull itself into a shape with the smallest amount of surface area. Surface tension is the attraction of water molecules to themselves helps to create a "film" on the surface of bodies of water. Small critters can "walk" across the water. Adhesion is when water is attracted to other materials.
States of Matter :
Water exists in 3 phases: Liquid (water), Solid (ice), and Gas (steam/water vapor). Temperature of a substance is a measure of the kinetic energy of the atoms and molecules which is measured in degrees (Celsius- science), Fahrenheit in US. Heat is a measure of the total kinetic energy of the atoms and molecules which are measured in calories. One calorie is equal to the amount of heat needed to raise the temperature of 1 gram of by 1 degree Celsius. Here is an example: below are two pictures. One picture is a pool and the other is a boiling pot.
Water exists in 3 phases: Liquid (water), Solid (ice), and Gas (steam/water vapor). Temperature of a substance is a measure of the kinetic energy of the atoms and molecules which is measured in degrees (Celsius- science), Fahrenheit in US. Heat is a measure of the total kinetic energy of the atoms and molecules which are measured in calories. One calorie is equal to the amount of heat needed to raise the temperature of 1 gram of by 1 degree Celsius. Here is an example: below are two pictures. One picture is a pool and the other is a boiling pot.

1. Which object has more heat?
- Pool has more heat because it's bigger than the pot.
2. Which object has a higher temperature?
-It is at boiling point then it must have higher temperature.
- Pool has more heat because it's bigger than the pot.
2. Which object has a higher temperature?
-It is at boiling point then it must have higher temperature.
Effects of Heat on Water : High heat of vaporization happens when a hydrogen bond must be broken, so water can absorb a lot of heat without changing phase. It evaporates slowly causing the cooling effect when you sweat. Water has to boil at a high temperature. Most of the water on Earth is a liquid which is good for the oceans. High latent heat of fusion happens when a lot of heat is released when water freezes and a lot of heat is absorbed when ice melts. Water temperature is more stable than air temperature and water moderate climate. When water has a specific heat, it can absorb high specific heat of water which helps regulate the rate at which air changes temperature, which is why the temperature change of seasons is so gradual, not sudden, especially near large bodies of water like the oceans.
Density is when the physical property of a substance that describes the degree of compactness of a substance or how closely packed together the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= mass/volume
How much stuff in how much space?
Usually substances becomes more dense (more compact) when they solidify. Water is different because water actually becomes less dense in its solid form. Water is the substance able to do this.
Density is when the physical property of a substance that describes the degree of compactness of a substance or how closely packed together the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= mass/volume
How much stuff in how much space?
Usually substances becomes more dense (more compact) when they solidify. Water is different because water actually becomes less dense in its solid form. Water is the substance able to do this.
DENSITY eXPERIMENT LAB
Materials:
1. Two 250ml beakers, one 500ml beaker
2. Electronic scale
3. Glass stirring rod
4. Pickling salt, water samples (warm & cold), and food coloring (blue and yellow)
5. An ipad to take pictures from the experiment.
1. Two 250ml beakers, one 500ml beaker
2. Electronic scale
3. Glass stirring rod
4. Pickling salt, water samples (warm & cold), and food coloring (blue and yellow)
5. An ipad to take pictures from the experiment.

Step 1: We mass each empty 250ml beaker. This is the record of the beakers
Beaker #1 Beaker #2:
112.9 grams 117 grams
Step 2: Measure 250ml of cold water and place into beaker #1 and 250ml of warm water into beaker #2 .
Beaker #1 Beaker #2:
112.9 grams 117 grams
Step 2: Measure 250ml of cold water and place into beaker #1 and 250ml of warm water into beaker #2 .

Step 3: To the Beaker #1, add 20 grams of salt and 5 drops of blue food coloring. Stir it until the salt has fully dissolved.
Step 4: Add 4 drops of yellow food coloring to the Beaker #2.
Step 4: Add 4 drops of yellow food coloring to the Beaker #2.

Step 5: Mass each beaker that included the water. This what i've got:
Beaker #1: 387.9g (rounded) Beaker #2: 358.9g (rounded)
Step 6: Calculate the mass, volume and density of the water in each beaker.
Beaker #1: Density = 387.9g/250ml = 1.55 g/ml
Beaker #2: Density = 358.9g/250ml = 1.43 g/ml
Beaker #1: 387.9g (rounded) Beaker #2: 358.9g (rounded)
Step 6: Calculate the mass, volume and density of the water in each beaker.
Beaker #1: Density = 387.9g/250ml = 1.55 g/ml
Beaker #2: Density = 358.9g/250ml = 1.43 g/ml
After you finished calculate the density. You can start doing PART 2. Pour Beaker #2 (the warm water) into the 500ml beaker and place the beaker on the table. Let the water settle until it has stopped moving. Now, take Beaker #1 (the cold water) and slowly pour the water into the 500ml beaker. Stop pouring once in a while and wait for the layers to settle, then you can continue pouring. Allow a minutes for the water to settle.

After that you can see the green water start separate out to two layers. Blue in the bottom and yellow is on top because they have different density. Out of this experiment I can see that thermal stratification is involved into this, it show you how important it is because by that you can bring nutrients and sources up to the land and oxygen into the water in order to fish and things that live under the sea to survive.
